产物官能团类型

1. LiAH4

羧酸在温和条件下可以通过氢化铝锂(LiAlH4)轻松还原。一个缺点是其缺乏选择性。四氢铝锂具有强烈的还原性,它会同时还原羧酸、醛、酮、酰胺、亚胺等不饱和基团[1]
案例
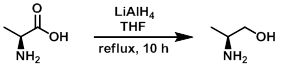
LiAlH4 (12.78 g, 336.7 mmol) was suspended in dry THF (550 mL) at 0 C. To this mixture was added the SM (15.0 g, 168.4 mmol) in portions over 20 min. The mixture was then refluxed for 10 h. After cooling the mixture to 0 C it was diluted with 2N NaOH (70 mL) and stirred at RT for 3 h. The resulting solids were filtered and washed with THF (200 mL). The solids were then suspended in THF (250 mL) and refluxed for 1 h. The solids were filtered and washed with THF (100 mL). The same trituration procedure was repeated twice and the resulting THF solutions (containing the product) were combined and used immediately in the next reaction.
Patent Reference: WO2012112946, page 43
2. BH3-THF

硼烷(BH3)试剂通常能够在酯的存在下选择性还原羧酸。硼烷-四氢呋喃(BH3-THF)的分解会生成氢气(H2)和三丁基硼酸酯。建议BH3-THF在低于35°C的条件下使用(出于安全考虑)。BH3-THF通常仅以1 M的浓度供应。[2][3][4]
案例
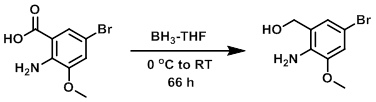
To a 0.24M THF suspension of the SM (71.7 mmol) at 0 C was added BH3-THF (1M, 220 mmol). The reaction mixture was stirred under Ar at RT for 66 h then quenched by addition of EtOH (15 mL) at 0 C and stirred an additional 15 min. The mixture was poured into H2O and extracted with DCM. The combined organics were washed with brine, dried (Na2SO4), and concentrated to provide the crude product as a white solid. [10.16 g, 62%]
Patent Reference: WO2007117607, page 308
3. BH3-Me2S
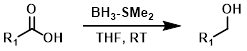
硼烷(BH3)试剂通常能够在酯的存在下选择性还原羧酸。硼烷-二甲基硫醚(BH3-SMe2)比硼烷-四氢呋喃(BH3-THF)更稳定,并且可以以更高的浓度(10 M)获得。BH3-SMe2的一个缺点是其难闻的气味。[2][3]
案例
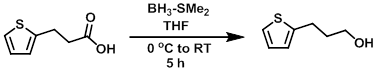
To a solution of the SM (1.0 g, 6.4 mmol) in THF (18 mL) at 0 C was added BH3-SMe2 (6.4 mL, 12.8 mmol). The reaction mixture was stirred at 0 C for 2 h, then RT for 3 h. The mixture was cooled to 0 C and quenched with sat aq K2CO3 (5 mL). The layers were separated and the aq layer was extracted with EtOAc (2 x 15 mL) and diethyl ether (2 x 15 mL). The combined org layers were dried (Na2SO4) and concentrated to provide the product as a colorless oil. [1.0 g, 100 %]
Patent Reference: WO2011061214, page 87
4. 活化硼氢化钠还原法
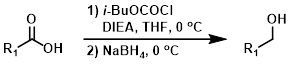
羧酸可以首先通过多种不同的试剂进行激活,然后用硼氢化钠(NaBH4)还原。当底物具有与LiAlH4或BH3不兼容的官能团时,这是一种有用的替代方法。
案例
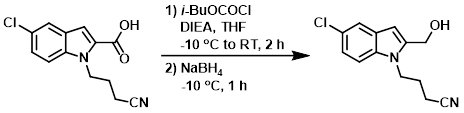
The SM (39.4 g, 149.98 mmol) and DIEA (51.69 mL, 300 mmol) were dissolved in THF (550 mL) and stirred at -10 C under N2. Then a solution of isobutylchloroformate in THF (50 mL) was added dropwise and stirring was continued for 1 h at -10 C, then 1 h at RT. NaBH4 (17.02 g, 450 mmol) was added portionwise at -10 C and stirred for 1 h. H2O (200 mL) was added cautiously to the reaction mixture and stirring was continued for another hour at RT under N2. The mixture was neutralized with 10% citric acid (aq) and then was extracted with EtOAc. The org layer was dried (MgSO4) and concentrated. The residue was purified by silica gel chromatography (50:50:0 to 0:100:0 to 0:99:1 heptane/DCM/MeOH) to provide the product as a white powder. [23.9 g, 64%]
Patent Reference: WO2015158653, page 35