产物官能团类型

--HATU-- --HBTU-- --EDC+HOBt-- --EDC+DMAP-- --DCC-- --T3P-- --BOP-- --PyBOP-- --CDI-- --酸酐法-- --琥珀酰亚胺酯法--
酰胺键的形成是一门成熟的化学,长期以来众多特质各异的试剂被开发出来,再次引入一篇综述(Chem. Soc. Rev., 2009,38, 606-631)来了解这门化学, 酰胺键的形成是有机合成中的一项基本反应,通常由多种所谓的偶联试剂介导。本综述重点介绍了最近开发的偶联试剂,特别关注基于首字母缩写的试剂的优缺点。其目的是揭开这一过程的神秘面纱,使化学家在进行酰胺偶联反应时能够做出合理且有根据的选择
1. HAUT
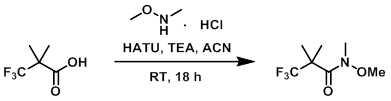
案例
To a solution of the acid (5.00 g, 32.0 mmol) in ACN (22.9 mL) was added TEA (9.82 mL, 70.5 mmol) followed by HATU (12.8 g, 33.6 mmol). The mixture was stirred at RT for 15 min, after which time the dark clear mixture was treated with the amine (3.44 g, 35.2 mmol). The reaction mixture was stirred at RT for 18 h. The mixture was diluted with EtOAc (100 mL) and washed with 1N HCl(2 x 100 mL), sat.NaCl (5 x 100 mL), dried Na2SO4), and concentrated. The resulting crude orange solid was adsorbed onto a plug of silica gel and purified by chromatography (0-25% EtOAc/heptane) to provide the product as a yellow liquid. [5.050 g, 79%]
Patent Reference: WO2015129926, page 124
反应机理
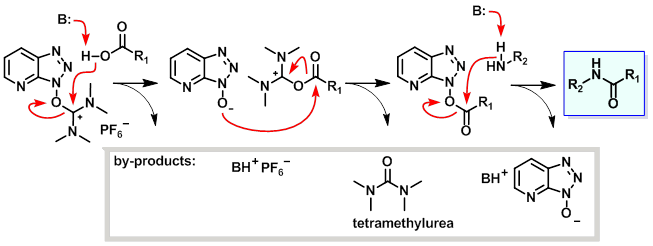
2.HBTU
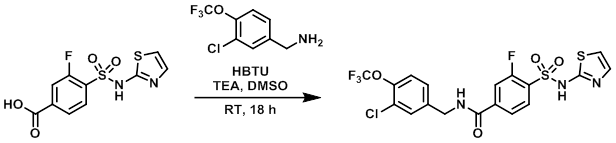
案例
To a suspension of the amine (3.0 g, 11.4 mmol), the acid (2.88 g, 9.54 mmol), and TEA (4.81 mL, 34.5 mmol) in DMSO (30 mL) was added HBTU (4.34 g, 34.5 mmol). The reaction mixture was stirred at RT for 18 h, after which time it was diluted with EtOAc (100 mL), washed with H2O (100 mL), brine (3 x 100 mL), dried (Na2SO4), and concentrated to a brown solid. The resulting material was triturated in MTBE then EtOAc to provide the product. [3.97 g, 7.8 mmol]
Patent Reference: WO2010035166, page 56
反应机理
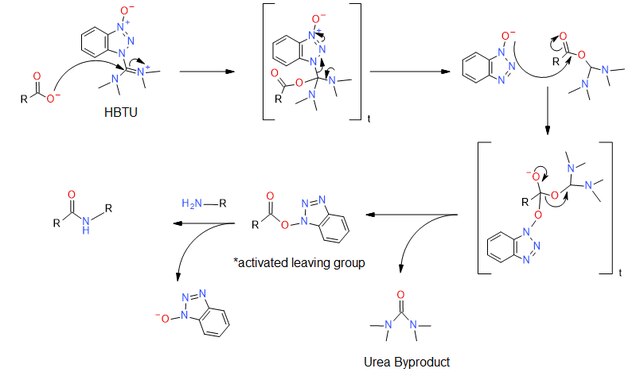
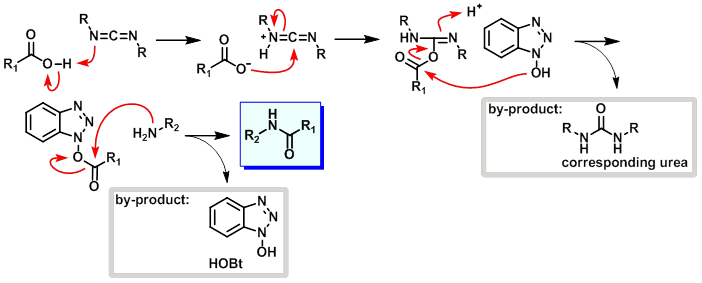
3. EDC+HOBt

案例
To a solution of the SM (1.0 g, 6.4 mmol) in DMF (5.0 mL) at 0 C was added EDC-HCl (1.2 g, 7.7 mmol), NH4Cl (1.4 g, 26.9 mmol), and HOBt (1.1 g, 8.3 mmol). The reaction mixture was stirred at RT for 12 h, after which time ice-cold H2O was added and the mixture was extracted with EtOAc. The org layer was dried (Na2SO4) and concentrated in vacuo to provide the product as an orange/red solid. [600 mg, 61%]
反应机理
4. EDC+DMAP
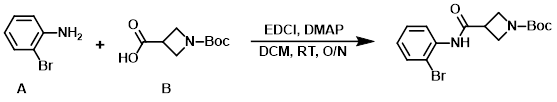
To a stirred solution of the amine (A) (150 g, 872 mmol) and DMAP (138.5 g, 1.133 mol) in DCM (2.5 L) was added the acid (B) (872 mmol) in one portion, followed by the addition of EDCI (217 g, 1.133 mol) in one portion at RT. The resulting mixture was stirred at RT overnight, after which time it was washed successively with 10% aq citric acid, H2O, sat aq Na2CO3, and brine. The org layer was dried (Na2SO4) and concentrated in vacuo to provide the product. [328 g, 85%]
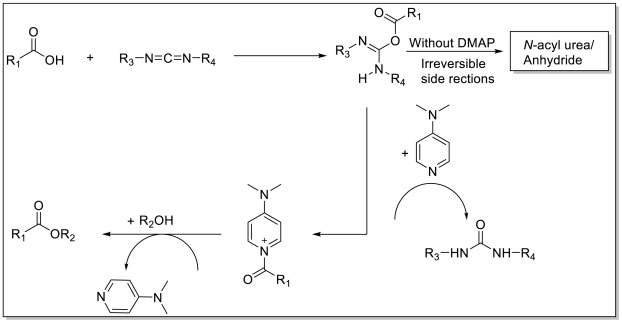
机理
5. DCC
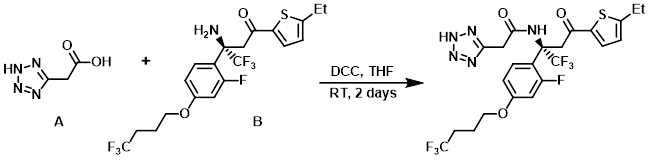
To a solution of the amine (B) (33 mg, 0.070 mmol) and the acid (A) (35.9 mg, 0.280 mmol) in THF (3 mL) at 0 C was added DCC (57.8 mg, 0.280 mmol). The resulting mixture was stirred at RT for 2 days. The solvent was removed in vacuo and the crude material was taken up in EtOAc and washed with sat aq NaHCO3, 1N HCl, and brine. The org layer was dried (MgSO4) and concentrated. The residue was purified by Prep HPLC to provide the product. [28 mg, 69%]
Patent Reference: WO2015191681, page 77
反应机理
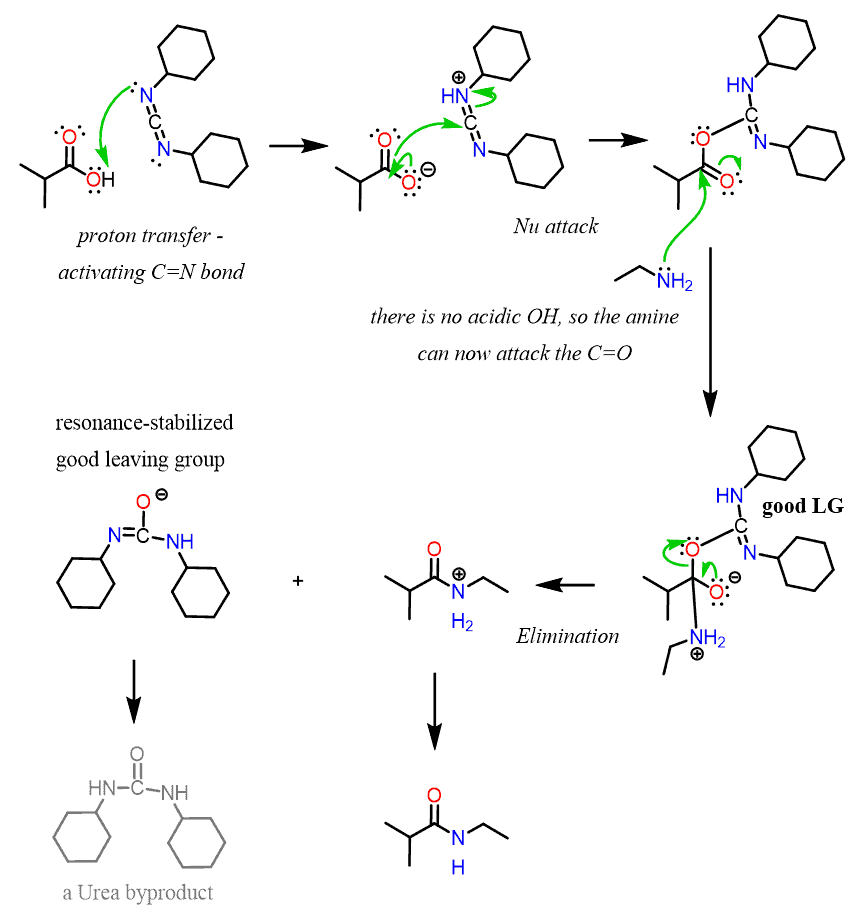
6. T3P

To a solution of the SM (2.92 g, 9.89 mmol) in 2-methyl tetrahydrofuran (115 mL) was added DIEA (13.8 mL, 39.5 mmol) and cyclopropylamine (2.77 mL, 39.5 mmol). The resulting mixture was heated to 70 C and T3P (50% weight solution in DCE) (17.3 mL, 59.3 mmol) was added. The reaction mixture was heated at 70 C for 16 h and then diluted with EtOAc (200 mL). The mixture was then washed sequentially with sat aq Na2CO3 (100 mL), 10% aq citric acid (100 mL), and saturated aq Na2CO3 (100 mL). The combined org extracts were dried (MgSO4) and concentrated in vacuo to give an oil. The crude was purified by column chromatography (eluting with 50% EtOAc/heptane) to give a solid which was recrystallized with IPA to provide the product as a white solid. [2.46 g, 74%]
Patent Reference: WO2010032200, page 94
反应机理
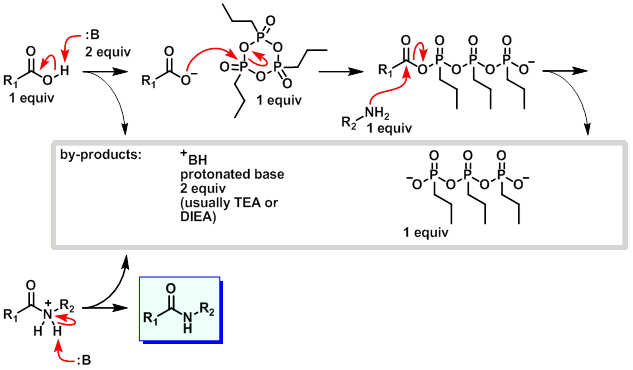
7. BOP
案例
To a mixture of the acid (0.3 g, 0.85 mmol) and BOP (442 mg, 1 mmol) in DMF at 0 C was added TEA (0.3 mL 2.14 mmol). The resulting mixture was stirred for 5 min, after which time the amine (110 mg, 1 mmol) in DMF was added dropwise and the reaction was stirred at RT for 5 h. The mixture was diluted with H2O and stirred overnight. The resulting solids were filtered and purified by chromatography (3% MeOH/CHCl3) to provide the product. [70 mg]
Patent Reference: WO2010038081, page 133
机理
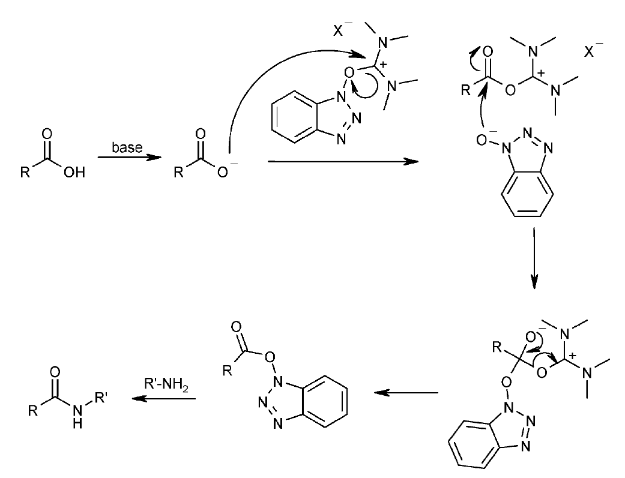
8. PyBOP
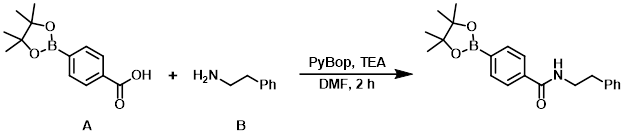
案例
A mixture of the acid (A) (200 mg, 0.81 mmol), the amine (B) (120 mg, 1.0 mmol), and TEA (0.29 mL, 1.6 mmol) in DMF (4 mL) was treated with PyBOP (380 mg, 1.0 mmol). After 2 h, the reaction mixture was concentrated in vacuo, suspended in H2O (25 mL), and stirred at RT. After 16 h, the mixture was filtered and the filter cake was washed with ether (2 x 2 mL) to provide the product as a yellow solid. [260 mg, 91%]
Patent Reference: WO2015105749, page 89
机理上,PyBOP是BOP的衍生试剂,在反应机理上具有类似性质。
9. CDI
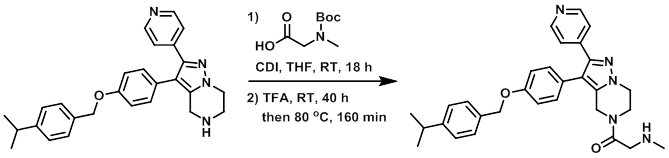
案例
The acid (152 mg, 0.801 mmol) was added to a solution of CDI (130 mg, 0.801 mmol) in dry THF (1 mL) at RT under N2. The mixture was stirred for 90 min at RT, after which time it was added over 25 min to a solution of the amine (200 mg, 0.400 mmol) in THF (1.5 mL) at RT under N2. The reaction mixture was stirred at RT for 16 h. Additional acid (152 mg, 0.801 mmol) and CDI (130 mg, 0.801 mmol) in dry THF (1 mL) was added slowly at RT. The reaction mixture was stirred at RT for 2 h. TFA (1 mL, 26.1 mmol) was added at RT, and the mixture was stirred 22 h. Additional TFA (1 mL, 26.1 mmol) was added, and the mixture was stirred 18 h. The reaction mixture was then stirred at 80 C for 160 min. The mixture was diluted with H2O and extracted with EtOAc. The org phase was washed with brine, dried (MgSO4), and concentrated to give an oil. The oil was purified by Prep LC (12 g silica, 100% DCM to 90:9:1 DCM/MeOH/NH3) to provide the product as a white solid. [158 mg, 80%]
Patent Reference: WO2015144799, page 287
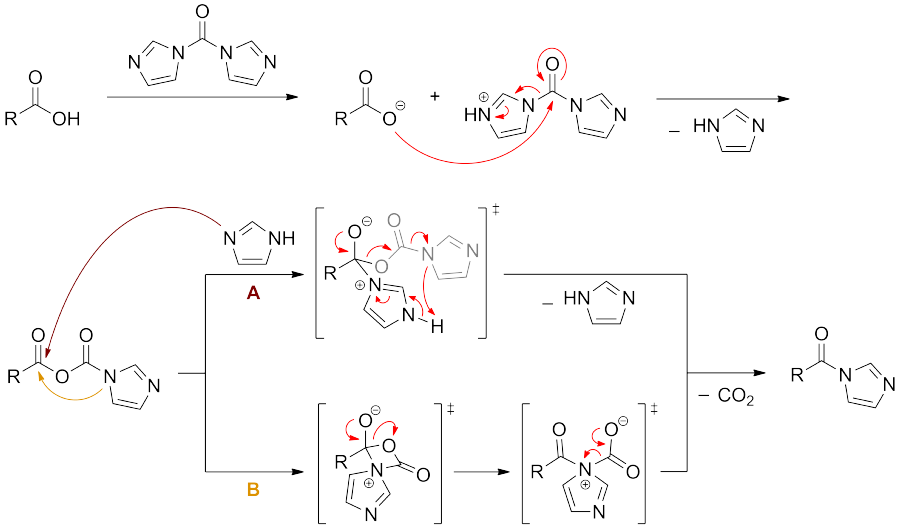
机理
酰胺的形成可通过碳二亚胺(CDI)促进。虽然 CDI 的反应性低于酰氯,但其操作更方便,并且避免了使用酰氯形成过程中可能引发副反应的亚硫酰氯(SOCl2)。 这种类型反应的早期应用可见于肽键的形成(以二氧化碳的生成作为驱动力)。以下为羧酸与 CDI 反应的推测机理:
10. 酸酐法
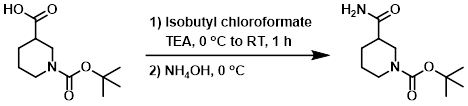
案例
To a solution of the SM (50.0 g, 218.0 mmol) in dry DCM (400 mL) at 0 C was added TEA (44.1 g, 437 mmol), followed by isobutyl chloroformate (596.0 g, 437.0 mmol). The reaction mixture was stirred at RT for 1 h, after which time it was concentrated in vacuo. The resulting material was cooled to 0 C and treated with NH4OH. The resulting solids were filtered, washed with H2O, and dried to provide the product as a white solid. [40.0 g, 80%]
Patent Reference: WO2014149164, page 213
11. 琥珀酰亚胺酯法
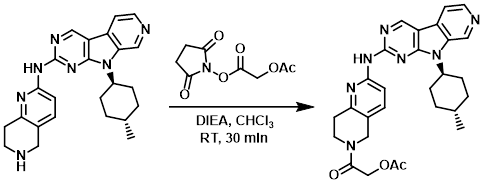
案例
To a suspension of the SM (827 mg, 2.0 mmol) in CHCl3 (10 mL) was added DIEA (0.348 mL, 2.0 mmol) and 2,5-dioxopyrrolidin-1-yl 2-acetoxyacetate (560 mg, 2.6 mmol). The reaction mixture was stirred at RT for 30 min leading to a yellow solution, after which time the reaction mixture was concentrated. MeOH (5 mL) and H2O (6 mL) were added to form a slurry which was stirred at RT for 1 h. The resulting solid was filtered to provide the product as a light yellow solid. [1.04 g, 98%]
Patent Reference: WO2012129344, page 132