产物官能团类型

1. Fisher酯化反应
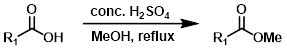
Fisher酯化反应最适合用于简单醇类(例如MeOH或EtOH),这些醇类可以大量过量使用(作为溶剂)。一元醇和二元醇反应效果良好,而三元醇和酚类效果较差。酸敏感底物可能无法很好地耐受。1
案例
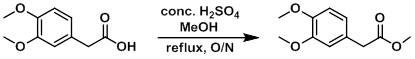
To a solution of the SM (25.0 g, 127.4 mmol) in MeOH (100 mL) was added a catalytic amount of H2SO4 (~10 drops). The mixture was refluxed overnight, after which time it was concentrated to remove MeOH and diluted with DCM (250 mL). The mixture was washed with H2O (5 x 20 mL), brine (20 mL), dried (Na2SO4), and concentrated in vacuo to provide the product as an orange oil. [25.77 g, 96%]
Patent Reference: WO2012112946, page 43
2. Steglich酯化反应(DCC+DMAP)
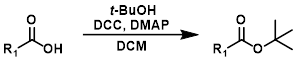
Steglich酯化反应是酸敏感底物的良好替代选择。EDC有时可替代DCC使用。该反应通常用于生成叔丁基酯。
案例
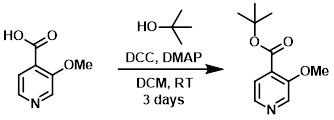
To a solution of the SM (10.0 g, 65.3 mmol) in DCM (400 mL) was added t-butanol (15.6 mL, 163 mmol), DCC (21.6 g, 104 mmol), and DMAP (16.0 g, 131 mmol). The reaction mixture was stirred at RT for 3 days. The solids were filtered away and the filtrate was concentrated in vacuo. The crude material was purified by silica gel flash chromatography, then triturated with 10% EtOAc/heptane. The solids were filtered away and the filtrate was concentrated in vacuo to provide the product. [9.7 g, 70%]
Patent Reference: WO2016014463, page 72
3. 酰氯甲醇一锅法
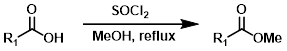
MeOH与SOCl2反应生成无水HCl,从而引发酸催化酯化反应(类似于Fischer酯化)。可以用EtOH代替MeOH来生成乙酯。酸敏感底物可能无法很好地适应此反应条件。[2]
案例

To a solution of the SM (1 g, 3.7 mmol) in MeOH (15 mL) was slowly added SOCl2 (5 mL) at 0 C. The reaction mixture was refluxed at 70 C for 2 h, after which time it was cooled to RT. MeOH was removed in vacuo and the resulting residue was poured onto ice-H2O (25 mL) and extracted with EtOAc (2 x 10 mL). The combined org extracts were washed with 10% NaHCO3 (2 x 10 mL), brine, dried (Na2SO4), and concentrated to provide the product as a yellow solid. [900 mg]
Patent Reference: WO2010038081, page 128
4. 酰氯法
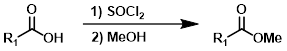
酯可以通过两步反应形成,首先用SOCl2(或草酰氯)生成并分离酸氯化物,然后与醇(此处为MeOH)反应生成酯。
案例
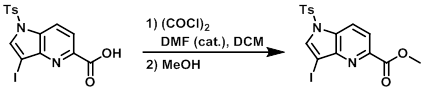
To a suspension of the SM (750 mg, 1.696 mmol) in DCM (8.5 mL) and a few drops of DMF was added oxalyl chloride (0.259 mL, 2.97 mmol) at RT. The mixture was stirred for 30 min, then cooled to 0 C and MeOH was added. The reaction mixture was stirred for 30 min, concentrated to a yellow solid, and diluted with H2O. The mixture was extracted with DCM (3x). The combined organics were dried (Na2SO4) and concentrated to provide the product as a pale yellow solid. [700 mg, 90%]
Patent Reference: WO2012129338, page 85
5. 碘甲烷
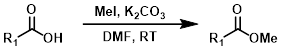
用碘甲烷(MeI)对羧酸进行烷基化可以生成甲酯。可以使用多种亲电试剂进行烷基化,这使其在某些情况下成为Fisher酯化的有用替代方法。一个缺点是可能会对其他亲核位点进行烷基化。[2]
案例
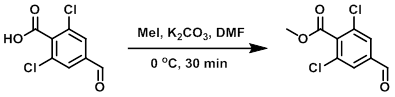
To a solution of the SM (1.1 g, 5.0 mmol) in DMF (10 mL) at 0 C was added K2CO3 (1.0 g, 7.5 mmol) followed by the slow addition of MeI (0.94 mL, 15.0 mmol), and the reaction was stirred at the same temperature for 30 min. The reaction mixture was quenched with H2O (50 mL) and extracted with EtOAc (2 x 30 mL). The combined organics were washed with H2O (50 mL), brine (50 mL), dried (Na2SO4), and concentrated. The resulting material was purified by silica gel column chromatography (10% EtOAc/hexane) to provide the product as a white solid. [0.59 g, 50%]
Patent Reference: WO2015129926, page 95
6. 硫酸二甲酯
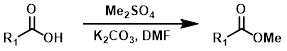
硫酸二甲酯是一种强效的甲基化试剂。当无法使用重氮甲烷或强酸性条件时,它是一种有用的替代方法。一个缺点是可能会对其他亲核位点进行甲基化。[4]
案例
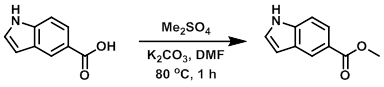
A solution of the SM (1.0 g, 6.2 mmol), K2CO3 (0.939 g, 6.8 mmol), and DMF (10 mL) was heated to 65 C and treated dropwise with Me2SO4 (0.78 g, 6.2 mmol) over 15 min, keeping the temperature between 65-70 C. The reaction was stirred at 80 C for 1 h, after which time it was poured into ice/water. The resulting solids were collected by filtration and dried in vacuo to provide the product. [0.91 g, 86%]
Patent Reference: WO2012129338, page 67
7.重氮甲烷法
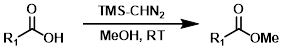
三甲基硅基重氮甲烷(TMS-CHN2)与羧酸快速反应生成甲酯。虽然在这些反应中推荐使用甲醇(MeOH),但甲醇并不是甲基化试剂。一个缺点是TMS-CHN2可能与其他位点(例如醇或酚)发生反应。[3] [4]
案例

Trimethylsilyldiazomethane (2.0M in hexane, 25 mL, 50 mmol) was added dropwise to a 0 C solution of the SM (4.64 g, 41.4 mmol) in DCM (25 mL) and MeOH (5 mL). The reaction mixture was stirred at RT for 30 min. The mixture was quenched with AcOH (0.45 mL) and concentrated in vacuo. The resulting material was purified by silica gel chromatography (20% DCM/hexane) to provide the product as a colorless oil. [3.8 g, 73%]
Patent Reference: WO2015129926, page 108