产物官能团类型
羧酸

1. Jones试剂/琼斯试剂
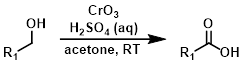
琼斯试剂是一种酸性的高价铬试剂,它不能兼容对酸敏感的官能团,而且产生具有高毒性的高价铬物种。该试剂 能够将伯醇氧化成酸,将仲醇氧化成酮。该方法应该尽量减少使用,尽可能探索可能的替代反应
案例
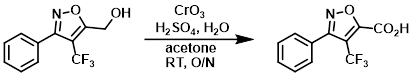
To an orange, homogeneous solution of CrO3 (12.4 g, 0.123 mol) in H2O (88.4 mL) at 0 °C was added H2SO4 H2SO4 (10.8 mL) dropwise via addition funnel over 30 min, with stirring. The addition funnel was rinsed with H2O (1 mL) to give a 1.23 M solution of Jones Reagent. To a solution of the SM (5.24 g, 21.6 mmol) in acetone (75 mL) at RT (immersed in H2O bath) was added Jones Reagent (43.8 mL, 53.9 mmol) via addition funnel over 90 min. The dark reaction mixture was stirred at RT overnight. By HPLC, the reaction was 93% complete. Additional Jones Reagent (18 mL, 1.0 equiv) was added. After stirring another 6.5 h, HPLC indicated 97% completion. Isopropanol (6 mL) was added, and the mixture stirred for 90 min, resulting in a dark green precipitate. The mixture was diluted with ether (600 mL) and washed with 2% aq NaHSO3 (5 x 100 mL). The layers were separated and the aq layer was back-extracted with ether (2 x 100 mL). The combined organics were washed with H2O (100 mL), brine (100 mL), and dried (Na2SO4). The aq layer was back-extacted with ether (100 mL), and the resulting org layer was combined with previous organics. The organics were concentrated to provide the pdt as an off-white solid. The pdt was dissolved in DCM (200 mL), washed with 2% aq NaHSO3, brine, dried (Na2SO4), and concentrated to provide the product (96% purity) as a pale yellow solid [3.84 g, 69.3%]. Additional product remained in the NaHSO3 aq layer. The aq layer was saturated with NaCl, the pH adjusted to ~3.5, and extracted with ether (3 x 100 mL). The organics were dried (Na2SO4) and concentrated to provide the product (99% purity) as a white solid [1.12 g, 20.2%]. Total yield: [4.96 g, 89.5%]
[Patent Reference: WO2011017578, page 69
CrO3 (cat.) + H5IO6
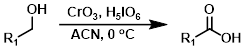
该方法是琼斯氧化的一种替代方法,使用催化量的CrO3和高碘酸来做氧化操作。该反应只需要 很少量的高毒性CrO3
案例
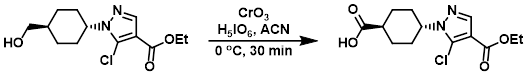
To a suspension of H5IO6 (159 mg, 0.698 mmol) in ACN was added CrO3 (0.6 mg, 0.0061 mmol), and the mixture was stirred at RT for 30 min. The mixture was cooled to 0 C and a solution of the SM (100 mg, 0.349 mmol) was added dropwise. The reaction mixture was stirred at 0 C for 30 min. The org solvent was removed in vacuo, the residue was supended in H2O (10 mL), and the mixture was extracted with EtOAc (3 x 10 mL). The combined organics were washed with H2O, dried (Na2SO4), and concentrated to provide the product as an off-white solid. [105 mg, quant.]
Patent Reference: WO2015129926, page 190
TEMPO氧化
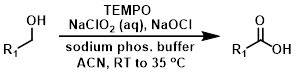
TEMPO氧化作为一种自由基氧化,是一种非常常用的氧化方式,它具有反应条件温和,反应选择性高的特点。作为催化剂 参与氧化反应时,可以用次氯酸钠,N-氯代丁内酰亚胺或其他氮的氯化物作为氧化剂,如TCCA。此处引用 Org. Process Res. Dev. 2010, 14, 1, 245–251.
案例
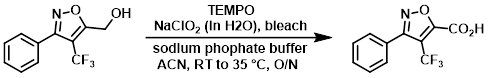
A fresh solution of sodium phosphate buffer (40 mL, pH ~6.5) consisting of a 1:1 solution of NaH2PO4 (20 mL, 0.67 M) and Na2HPO4 (20 mL, 0.67 M) was prepared. A mixture of the SM (2.1 g, 8.64 mmol), TEMPO (0.094 g, 0.604 mmol), and sodium phosphate buffer (32.2 mL, 21.59 mmol, 0.67 M) in ACN (30 mL) heated to 35 C. Solutions of NaClO2 (3.91 g, 34.5 mmol) in H2O (4.5 mL) and bleach (4.3 mL, 6% wt) were added simultaneously over 40 min. The reaction was monitored by HPLC. After 2 h, ~30% SM remained. After 6 h, ~10% SM remained. Additional bleach (100 uL) was added, and the reaction was left at RT overnight. Additional bleach (100 uL) was added. The resulting mixture was stirred at 35 C for 2 h, after which time HPLC indicated a complete conversion. The reaction mixture was quenched by the slow addition of solution of Na2SO3 (2.07 mL, 43.2 mmol) in H2O (90 mL) at 0 C, resulting in the disappearance of the brown reaction color. The solvent was removed in vacuo, and the remaining aq residue was extracted with EtOAc (3 x 40 mL). The organics were combined, washed with H2O (8 mL), brine (8 mL), dried (Na2SO4), and concentrated to provide the product as a pale yellow solid. [2.2 g, 99%]
Patent Reference: WO2011017578, page 71
NMO+TPAP(cat)
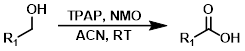
NMO的氧化和TEMPO氧化具有类似性,都是氮氧化物的氧化反应。此处反应用NMO作为当量的氧化剂,用TPAP (Tetrapropylammonium Perruthenate, 高钌酸四正丙基铵)作为催化剂。该反应具有非常好的官能团容忍性和选择性。
案例

To a solution of the SM (8.34 g, 18.15 mmol) in ACN (100 mL) was added NMO (8.51 g, 72.6 mmol) and TPAP (0.51 g, 1.45 mmol) in three portions. The resulting mixture was stirred at RT for 4 h. Then IPA (20 mL) was added and the mixture was stirred overnight. The mixture was concentrated in vacuo and the residue was partitioned between EtOAc (200 mL) and 1N HCl (80 mL). The layers were separated and the org layer was washed with brine, dried (Na2SO4), and concentrated to provide the product. [8.0 g, 16.90 mmol]