产物官能团类型

1.氟代反应
- DAST DAST试剂是一种亲核氟化试剂,该试剂与醇的反应是温和的,通常只需要0 °C到室温即可,非常活泼 的底物则需要-78 °C. 但是该试剂在加热(>90 °C)的情况下可能会发生分解,反应所需要的溶剂常常为DCM.
- Deoxo-Fluor Deoxo-Fluor也是一种亲核氟化试剂,但是往往比DAST有更好的活性,且具有更好的热稳定性。
案例1:DAST
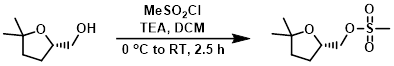
To a solution of the SM (3.3 g, 32.3 mmol) in DCM (100 mL) at -78 C under N2 was added DAST (13.0 mL, 96.9 mmol). The reaction mixture was stirred at -78 C for 1 h, then at RT for 1 h. The reaction mixture was then slowly poured into sat aq NaHCO3 (200 mL) at 0 °C. The mixture was extracted with DCM (2 x 60 mL). The combined organics were dried (Na2SO4) and concentrated. The residue was purified by silica gel column chromatography (10:1 PE/EtOAc) to provide the product as a colorless oil. [2.2 g, 65%]
Patent Reference: WO2016011390, page 382
案例2:Deoxo-Fluoro

The SM (50 mg, 0.135 mmol) was dissolved in anhydrous DCM (2 mL), and the mixture was cooled to 0 °C. Deoxo-Fluor (0.074 mL, 0.404 mmol) was added dropwise, the reaction mixture was stirred at 0 °C for 1 h, and then allowed to reach RT over 14 h. The mixture was quenched with MeOH (1 mL) and the solvent was removed in vacuo. The residue was purified by flash chromatography to provide the product as a white solid. [50 mg, 99%]
Patent Reference: WO2016010950, page 209
2.氯代反应
- 无溶剂二氯亚砜 醇可以在纯二甲亚砜中发生氯代反应,反应条件一般为回流~76 °C
- 有溶剂二氯亚砜 醇可以在二氯亚砜的溶剂中发生氯代反应,反应一般使用氯仿/二氯甲烷等溶剂。
- 二氯亚砜+溶剂+DMF DMF作为催化剂,可以与二甲亚砜形成活性中间体N,N-二甲基氯代甲基亚胺(Vilsmeier-Haack reagent),与 醇发生强烈的氯化反应。
- 磺酰氯 磺酰氯,例如甲磺酰氯/对甲苯璜酰氯,与醇发生温和的氯代反应。
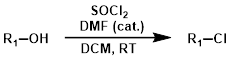
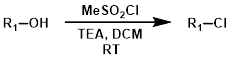
案例1:无溶剂二氯亚砜
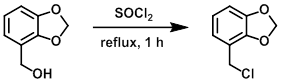
The SM (1.9 g, 12.5 mmol) was dissolved in SOCl2 (3 mL, 41.1 mmol) and refluxed for 1 h. The mixture was concentrated in vacuo to provide the product as a yellow oil that was used without further purification. [1.9 g, 91%]
案例2:二氯甲烷+二氯亚砜
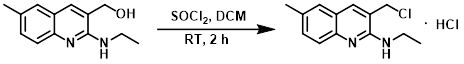
To a solution of the SM (460 mg, 2.13 mmol) in DCM (30 mL) was added SOCl2 (1.55 mL, 21.3 mmol). The mixture was stirred at RT for 2 h, after which time it was concentrated in vacuo. The residue was dissolved in DCM and concentrated twice to help remove any remaining SOCl2 and provide the HCl salt of the product as a yellow solid. [520 mg, 90 %]
Patent Reference: WO2005082859, page 145
案例3:二氯亚砜+DMF
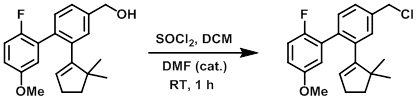
To a solution of the SM (1.10 g, 3.37 mmol) and a catalytic amount of DMF (0.10 mL) in DCM (12.0 mL) at 0 °C was slowly added SOCl2 (0.802 g, 6.74 mmol). The reaction mixture was stirred at RT for 1 h. The mixture was concentrated in vacuo and the resulting material was purified by silica gel column chromatography (eluting with 0-5% EtOAc/hexane) to provide the product. [1.15 g]
Patent Reference: WO2010045258, page 119
案例4:磺酰氯法
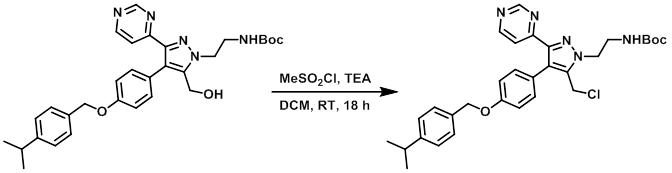
To a stirred solution of the SM (1.66 g, 3.05 mmol) in dry DCM (35 mL) at 0 °C was added TEA (0.679 mL, 4.89 mmol) and MeSO2 (0.354 mL, 4.58 mmol). The reaction mixture was stirred at RT for 18 h, after which time was added sat aq NaHCO3 and DCM. The org layer was separated, washed with brine, dried (MgSO4), and concentrated in vacuo to provide the product as a brown solid. [1.76 g, quant.]
Patent Reference: WO2015144799, page 325
3.溴代反应
- 三溴化磷(PBr3) 三溴化磷是最广泛使用的溴代试剂之一,其反应的主要机理是SN2
- Appel reaction Appel反应是烷基卤代的重要反应之一,其中溴代最常用的试剂是四溴化碳(CBr4) ,也可以使用其他溴源,例如Br2和NBS。 反应主要由三苯基膦和四溴化碳参与,生成三苯基氧膦作为主要的副产物,机理主要是SN2,涉及 构型的反转,且立体选择性较高。
- 二溴亚砜(SOBr2) 二溴亚砜具有极强的反应活性,在溴代反应中并不常见,且不能于吡啶共同使用,易形成不稳定复合物盐。
案例1:三溴化磷

To a solution of the SM (1.12 g, 6.66 mmol) in dry ether (19 mL) at 0 °C was added dropwise PBr3 (0.626 mL, 6.66 mmol). The ice bath was removed and the reaction was stirred for 3 h. H2O was carefully added to the mixture and the layers were separated. The org layer was washed with brine, dried (MgSO4), and concentrated in vacuo to provide the product. [1.49 g, 97%]
Patent Reference: WO2015144799, page 189
案例2-1:三苯基膦和四溴化碳
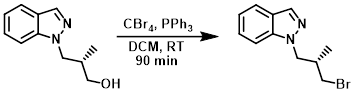
The SM (6.8 g, 0.036 mol) was taken up in DCM (275 mL) and treated with PPh3 (10.36 g) and CBr4 (13.1 g). The reaction mixture was stirred at RT for 90 min, after which time the solvent was concentrated to a volume of about 50 mL (precipitation occurs). The slurry was added to a pad of silica gel eluting with 20% EtOAc/hexane. Fractions containing the product (least polar) were combined and concentrated to provide the product as a viscous syrup. [6.2 g, 69%]
Patent Reference: WO2014152144, page 56
案例2-2:三苯基膦和Br2
To a solution of PPh3 (26.2 g, 0.10 mol) in ACN (50 mL) at 0 °C was added dropwise a solution of Br2 (5.13 mL, 0.10 mol) in ACN (30 mL). The SM (5.1 g, 0.050 mol) was added portionwise to the mixture and the reaction was stirred at 90 °C for 16 h. The mixture was concentrated and the resulting material was suspended in MTBE (150 mL). The solids were removed by filtration and the filtrate was concentrated. The resulting residue was dissolved in ACN and extracted with hexane (3 x 100 mL). The combined hexane extracts were concentrated in vacuo to provide the product as a brown oil. [6.5 g, 59%]
Patent Reference: WO2015129926, page 100
案例2-3:三苯基膦和NBS
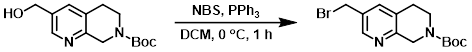
To a solution of the SM (0.261 g, 0.987 mmol) in DCM at 0 °C was added NBS (0.211 g, 1.19 mmol), followed by PPh3 (0.311 g, 1.19 mmol). The reaction mixture was stirred at 0 °C for 1 h, after which time it was concentrated in vacuo (without warming). The crude material was purified by silica gel flash chromatography to provide the product as a white solid. [0.22 g, 68%]
Patent Reference: WO2016014463, page 117
案例3:二溴亚砜 (SOBr2)
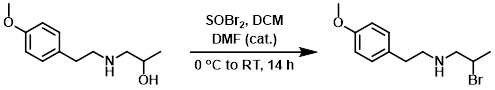
To a solution of the SM (2.63 g, 12.5 mmol) in DCM (60 mL) at 0 °C was added DMF (0.49 mL, 6.3 mmol) followed by SOBr2 (1.26 mL, 16.3 mmol). The mixture was allowed to warm to RT and stir for 14 h. Cold ether (30 mL) was added and the mixture was cooled to 0 °C, causing solids to precipitate out of solution. The solids were collected by filtration and dried on the filter pad to provide the product as an off-white solid. [3.31 g]
4.碘代反应
烷基醇的碘带反应的一个重要途径是appel反应。该反应在碘单质、三苯基膦和咪唑的参与下进行,主要遵从SN2的反应机理, 设计立体化学的反转
案例
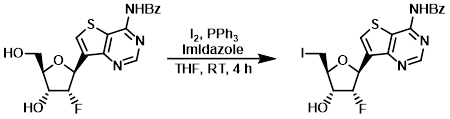
To a solution of the SM (0.79 g, 2.03 mmol), PPh3 (1.20 g, 4.58 mmol), and imidazole (0.277 g, 4.07 mmol) in THF (15 mL) was added iodine (0.96 g, 3.78 mmol) at RT. After 4 h, NaHCO3 (500 mg) was added to the mixture followed by H2O (200 uL) to quench the reaction. The mixture was concentrated and the residue was purified by silica gel column chromatography (1:1 EtOAc/hexane) to provide the product as a solid. [0.85 g, 84%]
Patent Reference: WO2016018697, page 79