产物官能团类型
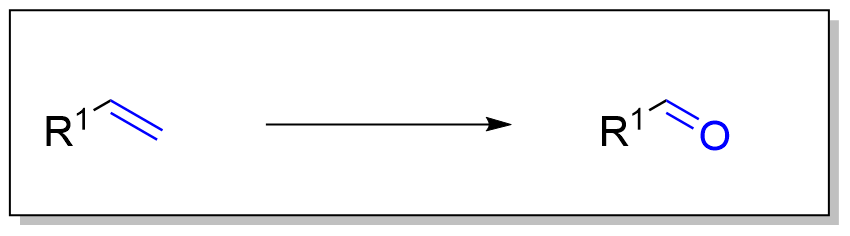
1. 臭氧化
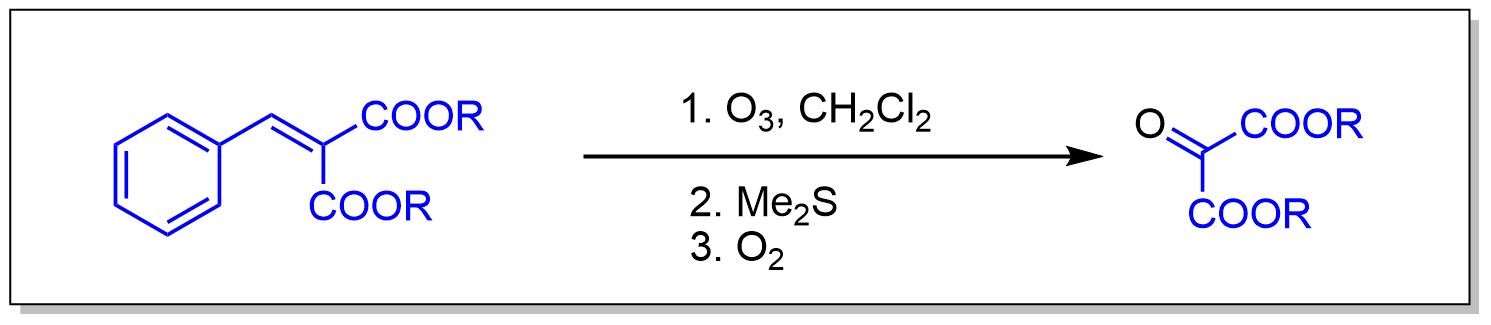
案例
A 300-mL wash bottle with an inlet tube fitted with a wide pore glass frit and equipped with a stirring bar is charged with 40.0 g (0.18 mol) of dimethyl benzalmalonate (Note 1) dissolved in 150 mL of dichloromethane. The cooled solution (0°C, ice bath) is purged with argon (10 min) and then a stream of ozone is passed through with vigorous stirring for 4.5 hr (Note 2).
After the reaction is complete (TLC, silica gel, diethyl ether/petroleum ether = 1:1), excess ozone is removed by purging with argon (10 min) and 15 mL of dimethyl sulfide (Note 3) is slowly added at 0°C (ice bath). Stirring is continued for 1 hr at this temperature and 2 hr at ambient temperature. Finally, air is blown through the solution for 12 hr (Note 4) and the residue is distilled at 20 mm, boiling range 90–100°C (Note 5) to give a yellow liquid that is further purified by filtration through 150 g of silica gel (SiO2) (Note 6) (elution with diethyl ether). The solvent is removed under reduced pressure and the residue is recrystallized from ethyl acetate to give 23.9 g (80%) of the dimethyl mesoxalate hydrate as colorless crystals (Note 7).
Dehydration of the product is accomplished by azeotropic removal of water. The hydrate is dissolved in dichloromethane (150 mL) and heated for 12 hr in a Soxhlet apparatus (Note 8) equipped with a thimble containing layers of phosphorus pentoxide and basic alumina. The solvent is then evaporated and the residue distilled at reduced pressure to give 20.1 g (76%) of the ester as a yellow liquid [bp 94°C (20 mm)].
四氧化锇(OsO4)
四氧化锇(OsO4)首先将烯烃氧化生成二醇,然后高碘酸钠(NaIO4)对其进行氧化性断裂。常用的溶剂体系包括 THF/H2O 或二氧六环/H2O。使用过量的 NaIO4 可再生 OsO4,因此仅需使用催化量的有毒 OsO4。[1]
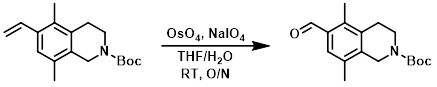
案例
The SM (1.03 g, 3.58 mmol), NaIO4 (2.34 g, 10.9 mmol), OsO4 (2.5 wt% in t-BuOH, 1.0 mL), THF (12.4 mL), and H2O (2.4 mL) were combined at RT. The reaction mixture was stirred overnight in the dark, after which time it was diluted with H2O and DCM. The layers were separated using a hydrophobic frit. The org layer was dried (Na2SO4) and concentrated. The residue was purified by silica gel flash chromatography to provide the product as an amber oil. [786 mg, 76%]
Patent Reference: WO2016014463, page 98