产物官能团类型
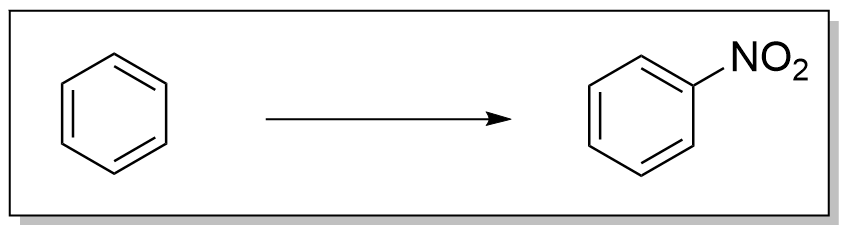
芳香化合物的典型硝化反应依赖于一种称为“混酸”的试剂,即浓硝酸和硫酸的混合物。 这种混酸生成硝酰阳离子(NO2+),这是芳香硝化反应中的活性物质。这种活性成分可以被分离,例如硝酰四氟硼酸盐(NO2+ BF4−)的情况下, 也可以在没有混酸的情况下进行硝化反应。在混酸合成中,硫酸不被消耗,因此既作为催化剂又作为水的吸收剂。在苯的硝化反应中,通常在不超过50°C的温和温度下进行。 该过程是亲电芳香取代反应的一个例子,涉及电子丰富的苯环的进攻:
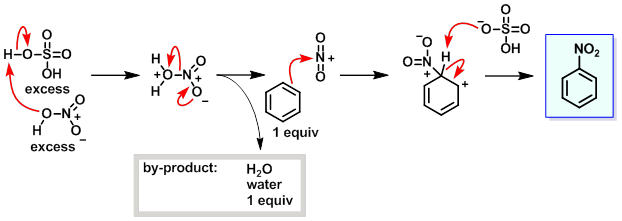
1. 硝酸-硫酸法
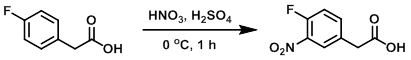
案例
To a solution of the SM (3.0 g, 19.46 mmol) in H2SO4 (20 mL) at 0 C was added dropwise HNO3 (0.913 mL, 20.44 mmol). The reaction was stirred at 0 C for 1 h. The mixture was poured onto ice and extracted with DCM (2x). The combined organics were washed with brine, dried (MgSO4), and concentrated to provide the product. [3.48 g, 90%]
Patent Reference: WO2013134298, page 35
2.醋酸-硝酸法
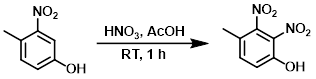
案例
To a solution of the SM (10 g, 65.36 mmol) in AcOH (60 mL) was slowly added HNO3 (3.3 mL, 78.4 mmol) at 0 C. The reaction mixture was stirred at RT for 1 h. The mixture was diluted with H2O (10 mL) and extracted with EtOAc (3 x 200 mL). The combined organics were dried (Na2SO4) and concentrated in vacuo. The residue was purified by silica gel column chromatography to provide the product as a light yellow solid. [5 g, 39%]
Patent Reference: WO2014149164, page 386