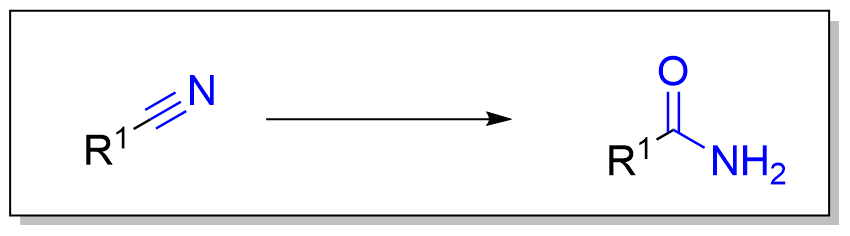
产物官能团类型
1. DIBAL-H

二异丁基铝氢化物 (DIBAL-H) 是将腈部分还原为醛的首选试剂。该反应通常通过将 DIBAL-H (~1 当量) 缓慢加入腈中并在低温(例如 -78°C)下进行。
案例
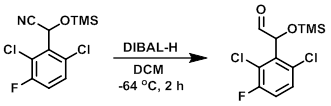
To a 100 mL, 3-necked, RBF was added the SM (0.50 g, 1.711 mmol) and DCM (9 mL). The mixture was purged with N2 and cooled to -64 C. Under N2 atmsophere, was added DIBAL-H (1.0M in hexane, 2.6 mL, 2.6 mmol). The mixture was stirred at -64 C for 2 h. While maintaining the temp below -65 C, the mixture was quenched with the careful dropwise addition of MeOH (1.4 mL, 34.2 mmol), followed by sat aq Rochelle salt (5 mL). The mixture was allowed to warm to RT and stir for 30 min. The mixture was diluted with H2O and DCM. The layers were separated and the aq layer was further extracted with DCM. The combined organics were washed with brine, dried (MgSO4), and concentrated to provide the product as a colorless oil. [0.517 g, crude]
Patent Reference: WO2015129926, page 111
2.SnCl2
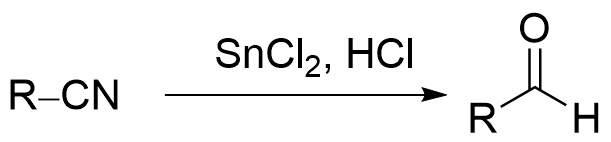
斯蒂芬醛合成是一种以化学家亨利·斯蒂芬(Henry Stephen,OBE/MBE)命名的化学反应。该反应通过使用氯化亚锡 (SnCl2) 和盐酸 (HCl),将腈 (R-CN) 转化为醛 (R-CHO)。反应过程中,生成的亚胺盐 ([R-CH=NH2]+Cl−) 需要用水 (H2O) 进行淬灭。[1][2]
案例
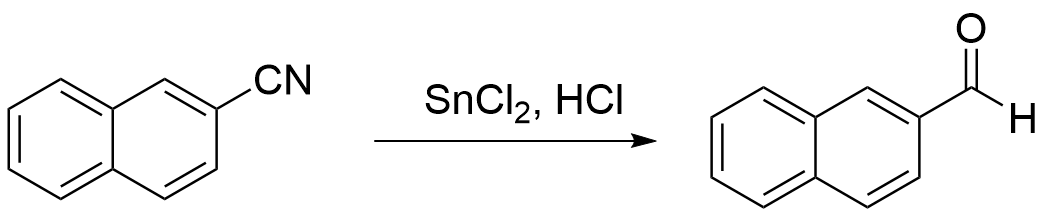
In a 2-l. three-necked round-bottomed flask, provided with a mechanical stirrer, a reflux condenser carrying a drying tube, and an inlet tube reaching nearly to the bottom of the flask, are placed 76 g. (0.4 mole) of anhydrous stannous chloride (Note 1) and 400 ml. of anhydrous ether. The mixture is then saturated with dry hydrogen chloride, while it is slowly stirred; this requires 2.5–3 hours, during which time the stannous chloride forms a viscous lower layer.
The inlet tube is replaced by a dropping funnel, and a solution of 30.6 g. (0.2 mole) of β-naphthonitrile, m.p. 60–62° (Note 2), in 200 ml. of dry ether is added rapidly. Hydrogen chloride is again passed into the mixture until it is saturated, and the mixture is then stirred rapidly for 1 hour and allowed to stand overnight while the yellow aldimine-stannichloride separates completely.
The ethereal solution is decanted, and the solid is rinsed with two 100-ml. portions of ether. The solid is transferred to a 5-l. flask fitted for steam distillation and immersed in an oil bath, the temperature of which is maintained at 110–120° (Note 3). Dry steam is passed through the mixture (Note 4) until the aldehyde is completely removed; this requires 8–10 hours, and 8–10 l. of distillate is collected. The white solid is filtered and allowed to dry in the air; it amounts to 23–25 g. (73–80%) and melts at 53–54°. For further purification, it is distilled under reduced pressure (Note 5); the water-clear distillate (b.p. 156–158°/15 mm.) is poured into a mortar while hot and is pulverized when cool. The recovery is 93–95%, and the melting point is 57–58°.
Org. Synth. 1971, 51, 20. 文献链接
3.HCOOH-雷尼镍

醛也可以通过使用氢供体随后对亚胺进行原位水解而生成。用于此反应的有效试剂包括在加氢催化剂存在下的甲酸[12],或用于向腈中添加一摩尔氢的金属氢化物。例如,硼氢化钠在酒精溶剂中通过 CoCl2 催化剂或雷尼镍(Raney nickel)可还原腈。
案例
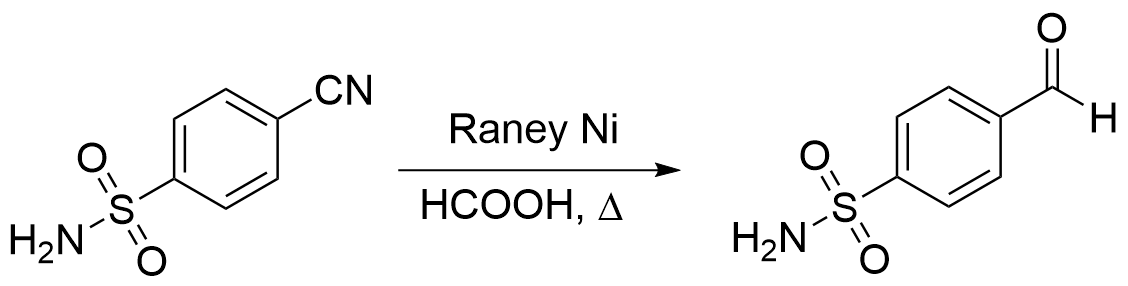
A 2-l., two-necked, round-bottomed flask fitted with a mechanical stirrer and a reflux condenser is charged with 40.0 g. (0.232 mole) of 4-cyanobenzenesulfonamide (Note 1), 600 ml. of 75% (v/v) formic acid, and 40 g. of Raney nickel alloy (Note 2). The stirred mixture is heated under reflux for 1 hour (Note 3). The mixture is filtered with suction through a Büchner funnel coated with a filter aid (Note 4), and the residue is washed with two 160-ml. portions of 95% ethanol. The combined filtrates are evaporated with a rotary evaporator (Note 5). The solid residue (Note 6) is dissolved in 400 ml. of boiling water and freed from a small amount of insoluble material by decantation through a plug of glass wool placed in a filter funnel. The filtrate is chilled in an ice bath, and the precipitate is collected by filtration with suction, washed with a small amount of cold water, and dried at 50° under vacuum, yielding about 32 g. of crude product, m.p. 112–114°.
The product is dissolved in 800 ml. of hot 95% ethanol, 15.5 g. of activated carbon (Note 7) is added, and the mixture is swirled periodically while it is allowed to cool for 1 hour. The activated carbon is removed by filtration with suction through a bed of filter aid (Note 4), the filter cake is washed with 50 ml. of 95% ethanol, and the combined filtrates are evaporated with a rotary evaporator. The residue is dissolved in 225 ml. of boiling water, and the hot solution is decanted through glass wool placed in a filter funnel. The filtrate is cooled to 0°, the product is collected by filtration with suction, washed with a small amount of cold water, and dried in a vacuum oven at 50°, yielding 25.6–28.0 g. (62.9–68.8%) of 4-formylbenzenesulfonamide, m.p. 117–118° (Note 8).
Org. Synth. 1971, 51, 20 文献链接