产物官能团类型
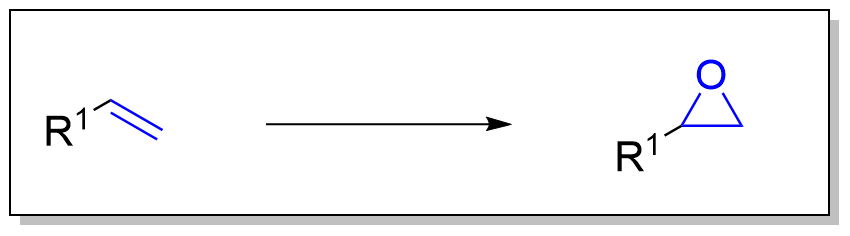
环氧化反应是有机化学中的基本反应,它将烯烃转化为环氧化物(氧杂环丙烷)。由于其高反应性和多功能性,环氧化物是有机合成中极为重要的中间体,可以进一步转化为多种功能团。 该反应通常涉及带有电亲氧的氧化剂,与双键反应生成三元环结构。
- 亲电加成:烯烃与氧供体(如过氧酸或二氧杂环丙烷)反应,形成环氧化物。
- 立体选择性:反应的立体选择性取决于反应条件和催化剂的选择。
方法总结
| 方法 | 试剂 | 特点 | 应用 |
|---|---|---|---|
| 过氧酸环氧化 | mCPBA、过乙酸、过邻苯二甲酸镁 | 简便、高效 | 将烯烃转化为环氧化物 |
| Sharpless环氧化 | 叔丁基过氧化氢(t-BuOOH)、Ti(OiPr)4、酒石酸酯 | 高对映选择性 | 手性丙烯醇的不对称环氧化 |
| Jacobsen环氧化 | Mn-salen络合物、漂白剂(次氯酸钠) | 广泛的底物范围 | 顺式取代烯烃的环氧化 |
| Shi环氧化 | 果糖衍生酮催化剂、过一硫酸钾(Oxone) | 无金属催化 | 多种烯烃的手性环氧化 |
| 金属催化环氧化 | 钼、钒或钌与过氧化氢或其他氧化剂 | 高效性和可扩展性 | 工业规模的环氧化 |
| 电亲性环氧化 | 卤代醇或其他电亲试剂 | 间接生成环氧化物 | 功能化底物的环氧化 |
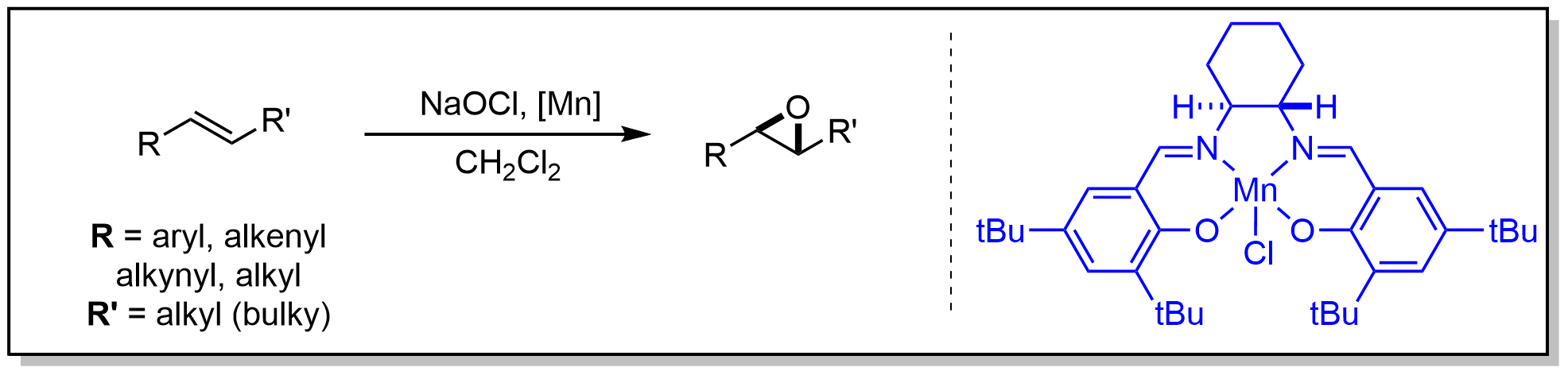
1. Jacobsen-Katsuki环氧化
Jacobsen环氧化反应通过使用手性锰-salen催化剂和化学计量氧化剂(NaOCl),能够从各种顺式取代的烯烃中选择性地生成环氧化物。与Sharpless环氧化反应相比,Jacobsen环氧化反应对底物的适应范围更广:良好的底物包括共轭顺式烯烃(R:芳基、烯基、炔基;R':甲基、烷基)或具有一个体积较大烷基基团的顺式烯烃。
案例
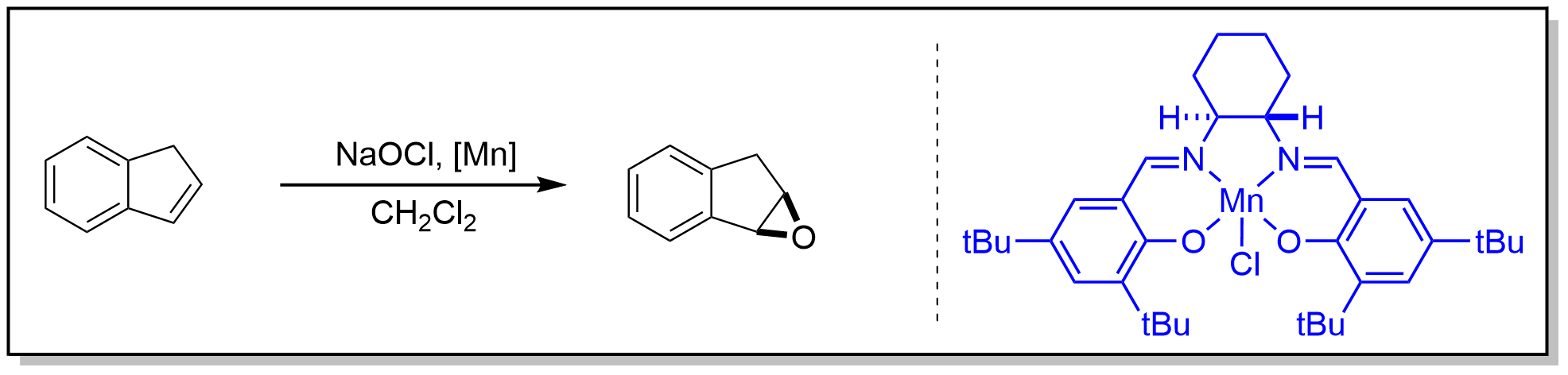
A 500-mL, three-necked, round-bottomed flask equipped with an overhead mechanical stirrer, a 125-mL addition funnel, and a thermocouple is charged with indene ((Note 1), 29.0 g, 0.25 mol, 1 equiv), dichloromethane (CH2Cl2) (30 mL), (S,S)-(N,N')-bis(3,5-di-tert-butylsalicylidene)-1,2-cyclohexanediaminomanganese(III) chloride (0.953 g, 1.5 mmol, 0.6 mol% , (Note 2)), and 4-phenylpyridine N-oxide ((Note 3), 1.28 g, 7.5 mmol, 3.0 mol%) under a nitrogen (N2) atmosphere. The resulting brown mixture is cooled to −5°C, and then a cold sodium hypochlorite (NaOCl) solution (191 mL, 1.7 M, 1.3 equiv, (Note 4)) is added slowly with vigorous stirring while maintaining the reaction temperature between 0°C and 2°C (Note 5). Upon complete addition of the bleach, the reaction is stirred for an additional 1 hr at 0°C. At this point, hexanes (200 mL) are added in one portion with stirring, and the reaction mixture is filtered through a pad of Celite on a large Büchner funnel. The filter cake is washed with dichloromethane (2 × 50 mL), and the filtrate is transferred to a 500-mL separatory funnel. The lower aqueous layer is removed, and the brown organic layer is washed with aqueous saturated sodium chloride (NaCl) solution (100 mL). The organic layer is dried over sodium sulfate (Na2SO4), filtered, and concentrated by rotary evaporation. A small amount of calcium hydride (CaH2) (100 mg) is added to the brown residue, and the epoxide is isolated by short path vacuum distillation, bp 58-60°C (0.025 mm), to yield 24.0 g of epoxyindane (84-86% ee) as a colorless to slightly yellow liquid (0.197 mol, 71% yield, (Note 6), (Note 7), and (Note 8)).
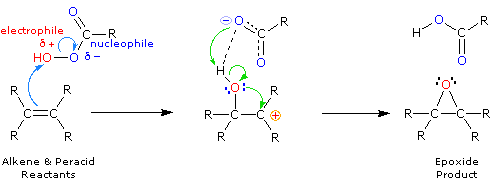
2.Prilezhaev环氧化

烯烃与过酸反应生成氧杂环丙烷(环氧化物)。商业上可获得的mCPBA(对氯过氧苯甲酸)是该转化中广泛使用的试剂,同时单过邻苯二甲酸镁和过乙酸也常被应用。
案例

A 250-mL, round-bottomed flask, equipped for magnetic stirring and with a condenser fitted with a calcium chloride drying tube, is charged with 9.76 g (40 mmol) of 1,1,1-trifluoro-2-ethoxy-5-phenyl-2-pentene , and a solution of 14.79 g (60 mmol) of 70% meta-chloroperoxybenzoic acid in 170 mL of dichloromethane (Note 10) and (Note 11). The resulting mixture is heated under reflux with stirring for 20 hr (Note 12), cooled, and concentrated to 50-60 mL under reduced pressure. The residual liquid is diluted with 200 mL of pentane and the supernatant liquid from the resulting suspension is passed through a short silica gel column (Note 8). The residual solids are washed twice with 50 mL of a 10:1 mixture (v/v) of pentane-diethyl ether , and the wash solutions are passed through the same column. The column is then eluted with 100 mL of a 5:1 (v/v) mixture of pentane-diethyl ether . The combined eluants are concentrated under reduced pressure and the residual liquid is purified by bulb-to-bulb vacuum distillation (oven temperature 120-130°C at 10 mm) to provide 9.36-9.88 g (90-95%) of pure 1,1,1-trifluoro-2-ethoxy-2,3-epoxy-5-phenylpentane (4) as a clear, colorless liquid, bp 90°C (10 mm)
3.Sharpless环氧化
Sharpless环氧化反应能够对前手性烯丙醇进行对映选择性环氧化。通过加入具有对映体过量的酒石酸衍生物实现不对称诱导。环氧化反应的氧化剂是过氧化叔丁醇。该反应由四异丙氧基钛 (Ti(OiPr)4) 催化,其通过氧原子与过氧化物、烯丙醇基团以及不对称酒石酸配体结合(假设的过渡态如下图所示)。
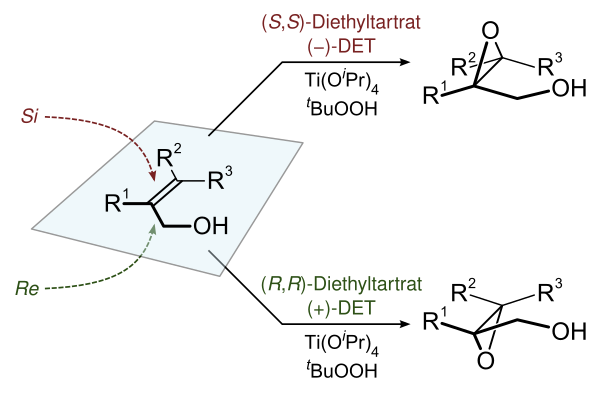
案例

A 500-mL, 1-neck round-bottom flask equipped with a Teflon-coated magnetic stir bar was oven dried and then fitted with a serum cap and flushed with nitrogen. The flask was charged with 200 mL of dry (distilled from CaH2 ) reagent-grade dichloromethane and cooled by stirring in a -23 °C bath Then the following liquids were added sequentially via syringe while stirring in the cooling bath: 5.94 mL (5.68 g, 20 mmol) of Ti(OiPr)4; 3.43 mL (4.12 g, 20 mmol ) of L(+)-diethyl tartrate (used as received from Aldrich), stirred 5 min before next addition; 3.47 mL (3.08 g, 20 mmol) of geraniol (Aldrich Gold Label); and, finally, ca. 11 mL of a dichloromethane solution (3.67 M in TBHP) containing ca. 40 mmol(2 equiv) of anhydrous tert-butyl hydroperoxide (TBHP).
The resulting homogeneous solution was then stored overnight (ca. 18 h) at ca. -20 °C in the sealed (serum cap) reaction vessel (the progress of the epoxidation can be monitored by TLC ) . Then the flask was placed in a -23 °C bath (dry and 50 mL of 10% aqueous tartaric acid solution was added while stirring; the aqueous layer solidified. After 30 min, the cooling bath was removed and stirring was continued at room temperature for 1 h or until the aqueous layer became clear. After separation of the aqueous layer, the organic layer was washed once with water, then dried over Na2SO4, and concentrated to afford a colorless oil with an odor revealing contamination by TBHP.
This oil was diluted with 150 mL of ether, and the resulting solution was cooled in an ice bath, and then 60 mL of 1 N sodium hydroxide solution was added. This produced a two-phase mixture which was stirred at 0 °C for 30min, The ether phase was washed with brine, dried over Na2SO4,and concentrated to give 4.24 g of a clear oil. Chromatography on silica gel afforded of 2(S),3(S)-epoxygeraniol (2.6 g, 77%), Analysis of this material as the "MTPA ester” gave an enantiomeric excess (ee) of >95% whereas analysis of the derived epoxy acetate by using Eu(hfbc)3 chiral shift reagent gave 94% ee.
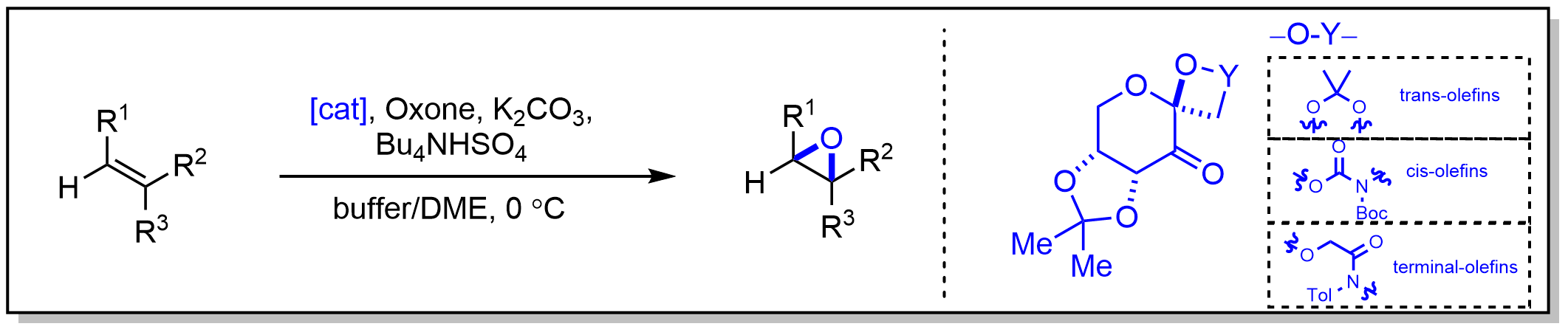
4.Shi环氧化
Shi环氧化反应通过使用一种来源于果糖的有机催化剂和 Oxone 作为主要氧化剂,从各种烯烃中合成环氧化物。 环氧化物的活性物质被认为是二氧杂环丙烷,它是一种强效的环氧化试剂。这些试剂并非无限稳定,但可以通过用过氧单硫酸钾(Oxone)氧化酮在原位生成。硫酸根作为一个良好的离去基团,促进了环的闭合生成二氧杂环丙烷。由于酮可以再生,因此只需要催化量的酮。此外,手性酮可以用于催化的不对称环氧化反应,因为酮的取代基接近反应中心。
该反应首次由史一安报道,其催化剂来源于D-果糖,具有一个靠近反应中心(酮)的立体中心。催化剂的刚性六元环结构以及相邻的季碳环基团最小化了该立体中心的差向异构化。由于对面 re-face 的位阻,活性二氧杂环丙烷催化剂从 si-face 进行氧化。该催化剂作为非官能化反式烯烃的不对称催化剂表现出高效性。
更多的关于史一安环氧化反应,请参见该Chemical Rev:Chem. Rev. 2008, 108, 9, 3958–3987
案例
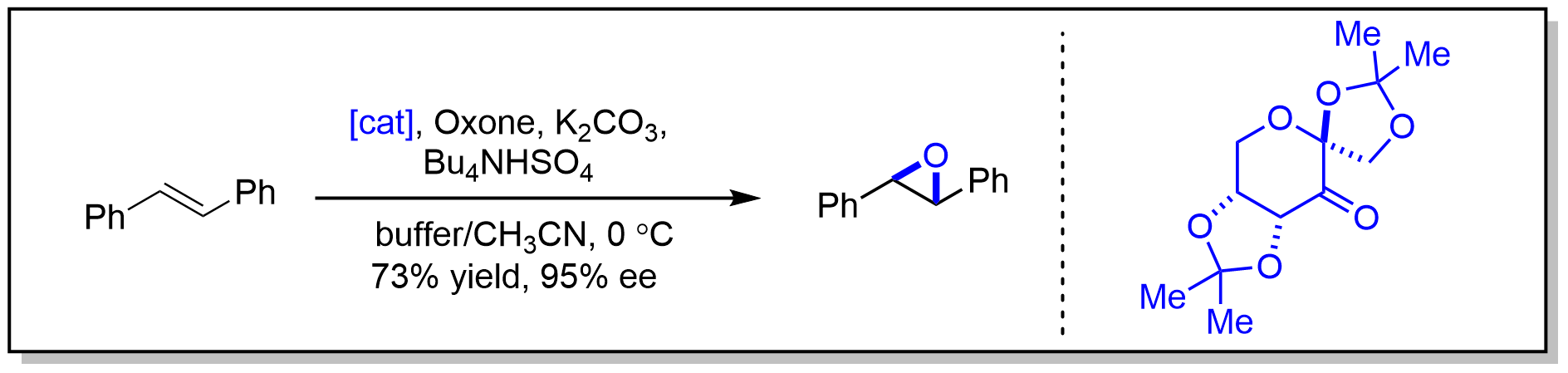
Aqueous Na2EDTA (1x10-4M, 10 mL) and a catalytic amount of tetrabutylammonium hydroxide were added to a solution of trans-stilbene (0.18 g, 1 mmol) in acetonitrile (15 mL) with vigorous stirring at 0 °C. A mixture of OxoneTM (3.07 g, 5 mmol) and sodium bicarbonate (1.3 g, 15.5 mmol) was pulverized. A small portion of this mixture was added to the reaction mixture to bring the pH to >7. After 5 min, fructose derived ketone (0.77 g, 3 mmol) was added portionwise over a period of 1 h. Simultaneously, the rest of OxoneTM and sodium bicarbonate was added portionwise over 50 min. After completion of the addition of ketone 3, the reaction mixture was stirred for another 1 h at 0 °C, diluted with water (30 mL) and extracted with n-hexane (4x40 mL). The combined extracts were washed with brine, dried (Na2SO4 ), filtered, concentrated, and purified by flash chromatography (the silica gel was buffered with 1% triethylamine solution in hexanes; hexanes/ether (1/0 to 50/1) was used as the eluent) to afford 0.149 g (73%) trans-stilbene oxide as white crystals.