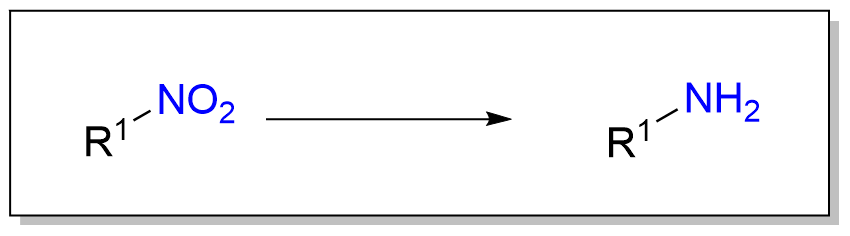
产物官能团类型
1. 钯碳氢化法
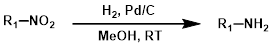
使用碳载钯(Pd/C)进行催化氢化通常是硝基还原的首选方法。芳香族和脂肪族硝基均可被还原为胺。然而,H2 + Pd/C 的一个缺点是它能够与底物上的多种其他官能团(氰基、卤素、烯键、炔键等)发生反应。
案例
10% Pd/C (2.50 g, 23.5 mmol) was added under a N2 atmosphere to a chilled (ice bath) solution of the SM (15.0 g, 67.5 mmol) in MeOH (400 mL). The flask was repeatedly evacuated and flushed with H2 gas. The resulting mixture was allowed to warm to RT and left to stir for 72 h under H2. The vessel was subsequently purged with N2 gas. The vessel was chilled (ice bath) and additional 10% Pd/C (2.50 g, 23.5 mmol) was added. The flask was repeatedly evacuated and flushed with H2. The mixture was allowed to warm to RT and stir for 6 h under H2. The vessel was purged with N2 and the crude reaction mixture was filtered through a short plug of celite. The reaction vessel and celite were rinsed with MeOH. The combined filtrates were concentrated in vacuo and the residue was dried under high vac overnight to provide the product as a blackish/grey solid. [12.1 g, 93%]
Patent Reference: WO2011014535, page 40
2.雷尼镍氢化法
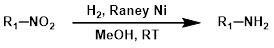
使用雷尼镍(Raney Nickel)进行催化氢化可以有效地还原硝基。在对芳香卤化物(I、Br 和 Cl)的脱卤反应可能引发问题的情况下,雷尼镍通常被用作 Pd/C 的替代品。[3]
案例

To a shaker flask was added the SM (crude, 32.4 g, 138 mmol), EtOH (100 mL), and Raney Ni (1.00 g, 17.04 mmol). The flask was charged with H2 (275 kPa) and was agitated until the absorption of H2 ceased. The vessel was depressurized and the catalyst was removed via filtration. The filtrate was concentrated to dryness, diluted with MTBE, and filtered again. The filtrate was concentrated and the resulting residue was stirred in hexane. The solids were filtered, washed with cold hexane, and dried in vacuo to provide the product as a dark solid. [17.8 g, 63%]
Patent Reference: WO2013134298, page 42
3.铁粉还原法
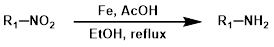
在酸性条件(例如醋酸,AcOH)下使用铁(Fe)为硝基还原为胺提供了一种温和的方法,即使存在其他可还原基团也能有效进行反应。
案例1

To a solution of the SM (70 g, 386 mmol) in AcOH (100 mL) and absolute EtOH (400 mL) was slowly added iron powder (40 g). The reaction was cooled in an ice-H2O bath and treated with conc. HCl (1 mL). The addition was exothermic. The reaction was heated to reflux for 20 min, after which time it was allowed to cool to room temperature. The mixture was filtered and the filtrate concentrated to a thick oil. The resulting oil was partitioned between EtOAc (500 mL) and H2O (200 mL) and basified to pH 10 using 6N NaOH. The mixture was filtered through celite. The org layer was separated and washed with sat aq NaHCO3 (2 x 100 mL), H2O (2 x 100 mL), brine (100 mL), dried (Na2SO4), and concentrated to provide the product. [37.2 g, 64%]
Patent Reference: WO2007117607, page 306
案例2
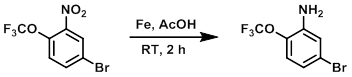
To a solution of the SM (2.0 g, 7.0 mmol) in AcOH (10 mL) at 0-10 C was added Fe powder (1.0 g, 17.9 mmol). The reaction mixture was allowed to stir at RT for 2 h. After completion, the reaction mixture was concentrated in vacuo and the resulting material was diluted with H2O and basified with sat aq NaHCO3. The mixture was extracted with EtOAc (2 x 50 mL), dried (Na2SO4), and concentrated to provide the product as a brown gummy solid. [1.2 g, 70%]
Patent Reference: WO2014149164, page 280
4.锌粉还原法
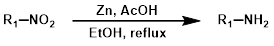
在酸性条件(例如醋酸,AcOH)下使用锌(Zn)为硝基还原为胺提供了一种温和的方法,即使存在其他可还原基团也能有效进行反应。
案例1
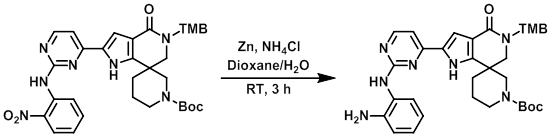
To a solution of the SM (130 mg, 0.2 mmol) in dioxane/H2O (3:1, 2 mL) was added NH4Cl (97 mg, 1.5 mmol) and Zn dust (80 mg, 1.5 mmol) at 0 C. The reaction mixture was stirred at RT for 3 h, after which time it was filtered through a celite bed. The filtrate was partitioned between H2O (10 mL) and EtOAc (30 mL). The org layer was separated, dried (Na2SO4), concentrated, and purified by column chromatography to provide the product as a yellow solid. [60 mg, 48%]
Patent Reference: WO2014149164, page 211
案例2
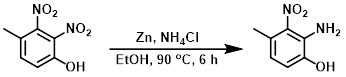
TTo a stirred solution of the SM (5 g, 25.25 mmol) in EtOH (60 mL) was added zinc powder (8.25 g, 126.26 mmol) and NH4Cl (6.8 g, 126.26 mmol). The resulting reaction mixture was stirred at 90 C for 6 h. The mixture was cooled to RT, filtered through a pad of celite, and concentrated in vacuo. The residue was purified by silica gel column chromatography to provide the product as a thick dark-brown solid. [2.5 g, 59%]
Patent Reference: WO2014149164, page 387
5.SnCl2还原法
使用二氯化锡(SnCl2)提供了一种温和的方法,可在存在其他可还原基团的情况下将硝基还原为胺。
案例1
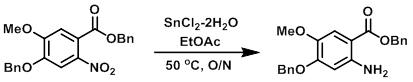
To a solution of the SM (10.9 g, 27.7 mmol) in EtOAc (100 mL) was added SnCl2-2H2O (18.7 g, 83.1 mmol). The reaction mixture was stirred at 50 C overnight. The mixture was filtered through celite, and the filtrate was washed with 10% NaHCO3. The layers were separated and the aq layer was further extracted with EtOAc. The combined organics were dried and concentrated in vacuo to provide the product as a brown solid. [9.5 g, 95%]
Patent Reference: WO2002016361, page 35
案例2
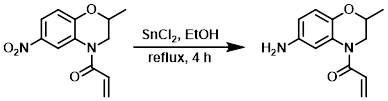
To a solution of the SM (700 mg, 2.81 mmol) in EtOH (5 mL) was added SnCl2 (2.8 g, 14.7 mmol) at 0 C. The resulting mixture was stirred at RT for 10 min, then reflux for 4 h. After completion of the reaction, ice-cold H2O was added to the reaction mixture. The residue obtained was diluted with 20% NaOH and the aq layer was extracted with EtOAc. The org layer was dried (Na2SO4) and concentrated to provide the product as a brown oily liquid. [560 mg, 91%]
Patent Reference: WO2014149164, page 375
6.Na2S还原法

硫化钠(Na2S)可用于在氢化或酸性条件不兼容的情况下处理底物。Na2S有时可以选择性地还原一个硝基,而其他硝基保持不变。通常,Na2S不还原脂肪族硝基基团。
案例1
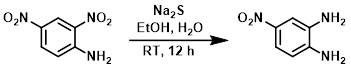
To a solution of the SM (1 g, 10.3 mmol) in 3:1 EtOH/H2O (26 mL) was added Na2S (1.2 g, 15.4 mmol). The reaction mixture was stirred at RT for 12 h. After completion of the reaction, solvent was removed in vacuo. The resulting residue was diluted with H2O and extracted with EtOAc. The org layer was dried (Na2SO4) and concentrated. The resulting material was purified by silica gel column chromatography to provide the product as a brown solid. [200 mg, 24%]
Patent Reference: WO2014149164, page 480
7.LiAlH4还原法
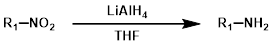
LiAlH4可将脂肪族硝基化合物还原为胺,但芳香族硝基化合物会生成偶氮化合物。LiAlH4是还原通过亨利反应(Henry reaction)生成的硝基烯烃的常用试剂。[2][3]
案例1
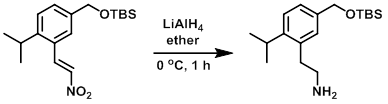
LiAlH4 (283 mg, 7.45 mmol) was added to a stirred solution of the SM (1.0 g, 2.98 mmol) in ether (30 mL) at 0 C. The reaction mixture was stirred at 0 C for 1 h. To the mixture was then added H20 (0.280 mL), 3N NaOH (0.280 mL), and H2O (0.840 mL). The resulting precipitate was removed by filtration and the filtrate was concentrated in vacuo to give a yellow oil. The oil (0.9 g) was purified by Prep LC [50 g silica, 0-20% (10% NH3 in MeOH)/(DCM)] to provide to product as a colorless oil. [250 mg]
Patent Reference: WO2015144799, page 341
Comments: